The Food and Drug Administration issued a warning about the dangers of using unauthorized versions of fat-dissolving injections, saying it has received reports of severe side effects, such as persistent scarring, severe infections and skin deformities.
Fat-dissolving injections, sometimes called lipolysis injections, are nonsurgical procedures that aim to melt away small amounts of fat beneath the skin. These injections are typically administered in so-called problem areas such as the chin, legs, upper arms and abdomen.
The FDA has approved one such injection, a drug called Kybella, from Kythera Biopharmaceuticals.
However, the FDA on Wednesday noted the presence of unapproved fat-dissolving injections popping up at clinics and med spas across the U.S., including those sold online under brand names like Aqualyx, Lipodissolve, Lipo Lab and Kabelline.
Common ingredients in the injections include phosphatidylcholine and sodium deoxycholate, two chemicals that have not been approved for use by the FDA.
The FDA said it received reports of consumers experiencing serious side effects following the use of unapproved fat-dissolving injections, including:
- Permanent scars
- Serious infections
- Skin deformities
- Cysts
- Painful knots
In some cases, the agency said, the injections were administered at clinics or med spas by individuals possibly lacking proper licensing.
Dr. Dan Maselli, an obesity medicine physician at True You Weight Loss, a weight loss clinic in Atlanta, said he’s also heard of people injecting themselves with fat-dissolving products they purchased online.
Maselli noted that carries considerable risk.
Aside from not knowing what chemicals are used in the product, the “patient might not know what they’re injecting into,” he said.
“They might think it’s fat tissue,” he said. “It could be a muscle tissue. It could be your blood vessel, or it could be around a nerve.”
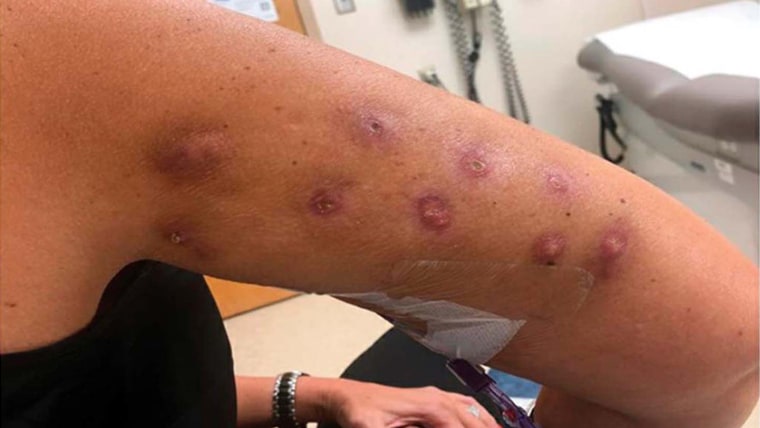
In the release, the FDA highlighted one woman who developed knots on both of her arms one week after receiving unapproved injections.
Dr. Shauna Levy, a specialist in obesity medicine and the medical director of the Tulane Bariatric Center in New Orleans, said people often seek these kinds of treatments as a quick fix to their weight loss goals or for cosmetic reasons.
“Especially this time of year,” she said. “People have holiday parties and they want to look up to society’s standards.”
However, she added, the treatments don’t get to the root of the problem, which is often more about a person’s metabolic health. GLP-1 medications, like Ozempic and Wegovy, and bariatric surgery, she said, aim to improve a person’s metabolic health.
“It’s just going to have the fat die,” Levy said of the fat-dissolving injections. “And I could totally see how people are having these types of reactions. Because when the fat just dies, the tissue’s going react like that.”
Maselli said if people are seeking these kinds of injections for more cosmetic reasons, rather than weight loss, he recommended seeing a licensed plastic surgeon.
The FDA also urged consumers to avoid purchasing fat-dissolving products from websites, noting that the products might be ineffective and may carry a risk of severe side effects.
If a person is experiencing side effects from the unapproved injections, the agency recommended seeing a health care provider.






